Hydrocracking is a process which eliminates aromatics and polar compounds from base stocks. This is achieved by chemically reacting the base oil with hydrogen in the presence of a special catalyst at very high temperatures and pressures.
During this process there are numerous reactions occurring, some being:
- Removal of polar compounds which contain sulphur, oxygen and nitrogen.
- Conversion of aromatic hydrocarbons to saturated cyclic hydrocarbons.
- Breaking down of heavy polycyclo-paraffins to lighter saturated hydrocarbons
The reactions that take place during this process happen under temperatures as high as 537ºC, pressures around 3,000 psi and in the presence of a special catalyst. The hydrocarbons which are formed as a result of this are very stable which makes them ideal for use as lubricant base oils.
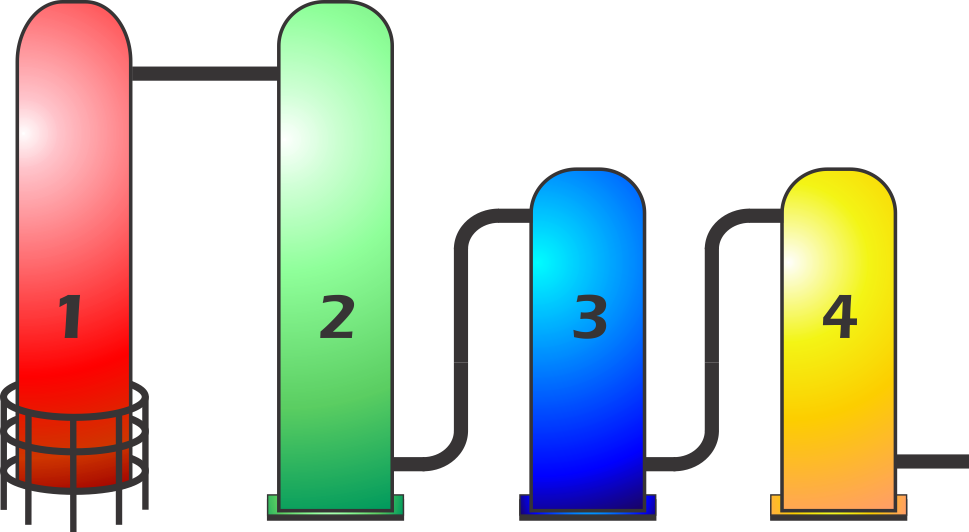
There are four stages in Hydrocracking.
1. Initial oil products are distilled in a special vacuum distillation column which are then divided into distillates and vacuum gas-oils.
2. Waxy lube base oils are moved into the hydrocracking chamber, under extreme pressure and temperatures the molecules become chemically active. Hydrogen is added to remove polar compounds and convert aromatic hydrocarbons into saturated cyclic hydrocarbons.
3. These are then moved to another hydrocracking chamber for additional hydrogen saturation, breaking down the heavy polycyclo-paraffins at the molecular level into lighter saturated hydrocarbons. This ensures that the oil is easily pumped at low temperatures.
4. The final hydrocracking chamber removes the last traces of aromatic hydrocarbons and polar compounds. This stabilizes the molecular structure of the base oil to ensure a superior resistance to oxidation which results in a longer life for the lubricant.
The end result is a base oil as clear as water. Hydrocracked base oils also offer other attractive features as lubricating oils such as:
- High Thermal Stability
- Superior Oxidation Resistance
- Very High Viscosity Index
- Outstanding Low Temperature Fluidity



